QMS Document Control Software FAQ’s
What is document control software?
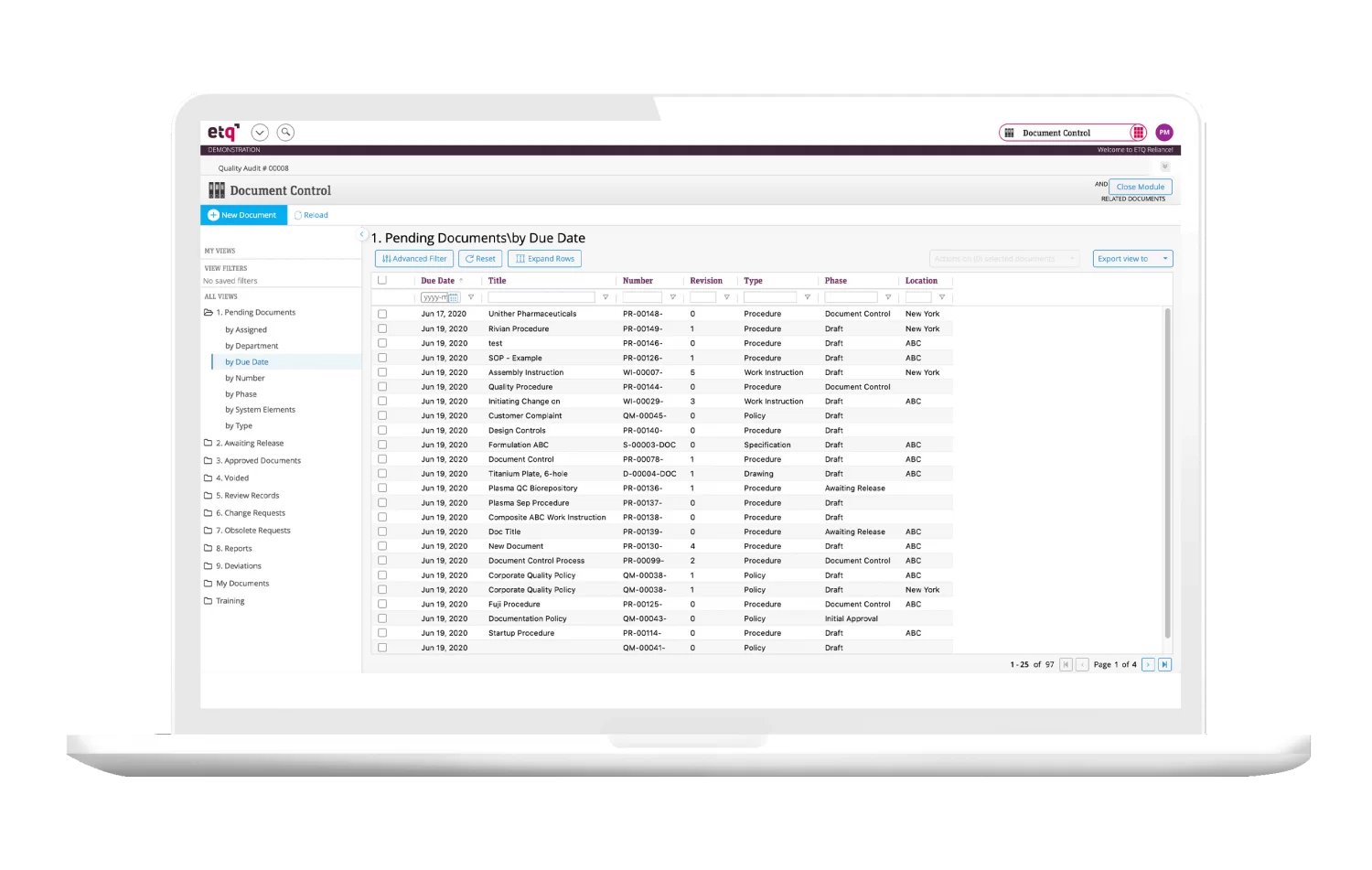
Document control software involves sorting and tagging production, manufacturing, regulation, and quality documents. This system allows users to immediately retrieve stored documents related to a specific topic instead of hunting through email inboxes and file trees.
How does a documentation control system work?
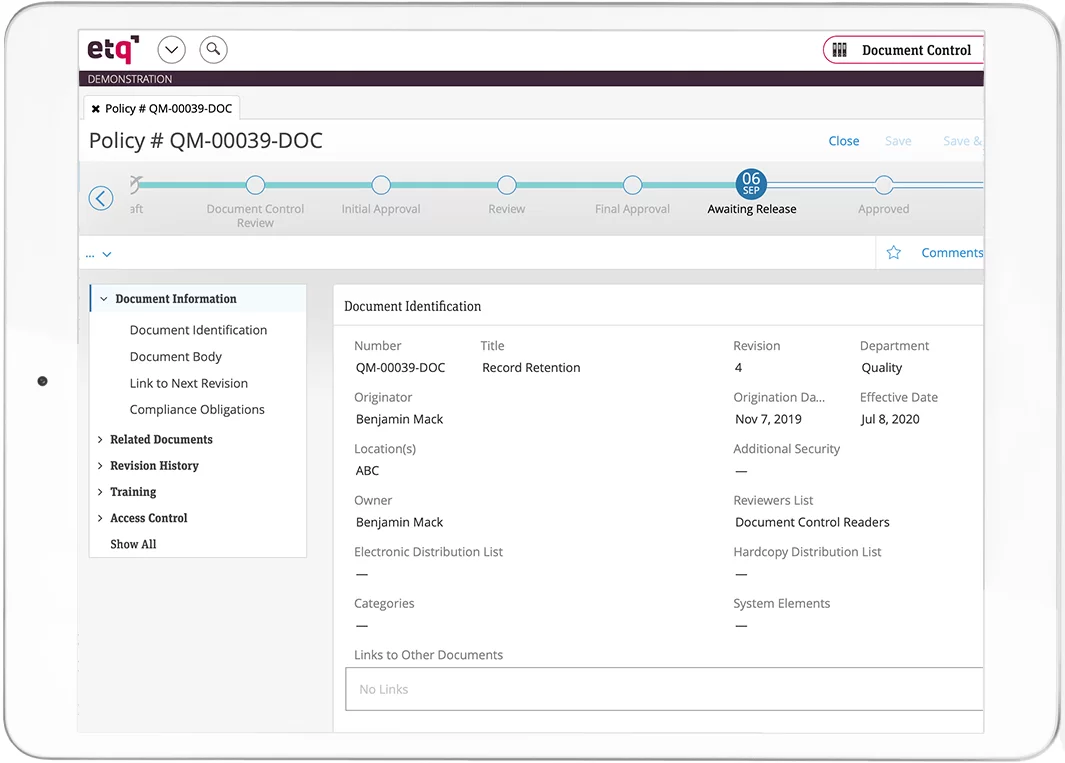
A document control system works by collecting metadata related to document creation. A training document, for example, would aggregate information on who created it, who checked and approved the document, its approval status, and who has the authorization to view, edit, or delete it. This feature makes it easy for users to find the document and replace it with a newer version when needed.
What are the advanced features of document control tools?
Document control tools may have several key features. The first is access control—the ability to control document access based on a user’s role within an organization. Automation is another advanced feature. You could use this to send the correct training documents to new hires automatically once they’re onboarded. Last, collaboration features let a group of employees complete a review and approval process while sidestepping version control issues.
How can a document control system improve compliance and efficiency?
Document control systems make it easy to store and retrieve documents. For example, if you’re under an audit, you’ll be able to retrieve documents related to the process under audit immediately. Since you don’t have to search for those documents manually, you’ll save hours—plus, there’s no risk of accidentally leaving anything out.
What are the benefits of documentation control software?
A significant benefit of documentation control software is the ability to pull and replace all out-of-date materials automatically. This benefit means that there’s little risk of a worker accidentally using an outdated production manual, which would risk interrupting critical business processes. In short, documentation control directly translates into increased worker safety and productivity.
How does document control improve management productivity?
Document control improves productivity in several respects:
- It saves hours searching for documents.
- It streamlines the review and approval process, speeding up document creation.
- It ensures that workers use standardized processes and safety procedures, which accelerates production on the factory floor.
What type of QMS document control system software do I need?
The QMS document control system software you’ll need largely depends on the industry in which you’re involved. The more tightly regulated your industry, the more robust document storage system you’ll need to collect and recover documents. For example, life science companies might need a more extensive document control system than a CPG company.
Is this software easy to use?
Yes, especially relative to the tasks that it’s replacing. It might take hours or days to sort through databases, file shares, and email threads to find the documents you need. Using document control allows retrieving those documents in minutes while minimizing the risk of mistakes.
Does document control system software integrate with other technologies?
Document control software interfaces with other QMS applications and external applications, like CRM, ERP, and ServiceDesk. This integration allows it to capture quality information created elsewhere in the company and sort it for later use.
Is document control software secure?
Documentation control software frequently uses a security method known as role-based access control (RBAC). With this method, accessing, modifying, or deleting a document is based on a user’s role within the company. An ordinary line worker probably wouldn’t be capable of modifying a training document, but their supervisor might. This feature helps keep sensitive documents secure and accurate, preventing privacy violations and increasing worker safety.
Which software is best for document control?
ETQ Reliance NXG cloud-based solution contains a robust document control application with powerful automation, security, version control, and more features. With Reliance NXG, quality managers can use document control as a single source of truth for critical data, ensuring that critical business documentation is kept up to date and never lost.
What software packages do Document Controllers use?
Document control applications are used most often in conjunction with quality management systems (QMS), enterprise resource planning (ERP), customer relationship management CRM), and ServiceDesk software. Because these applications produce a large amount of important information related to compliance and operations, document control must integrate with them and capture their output.