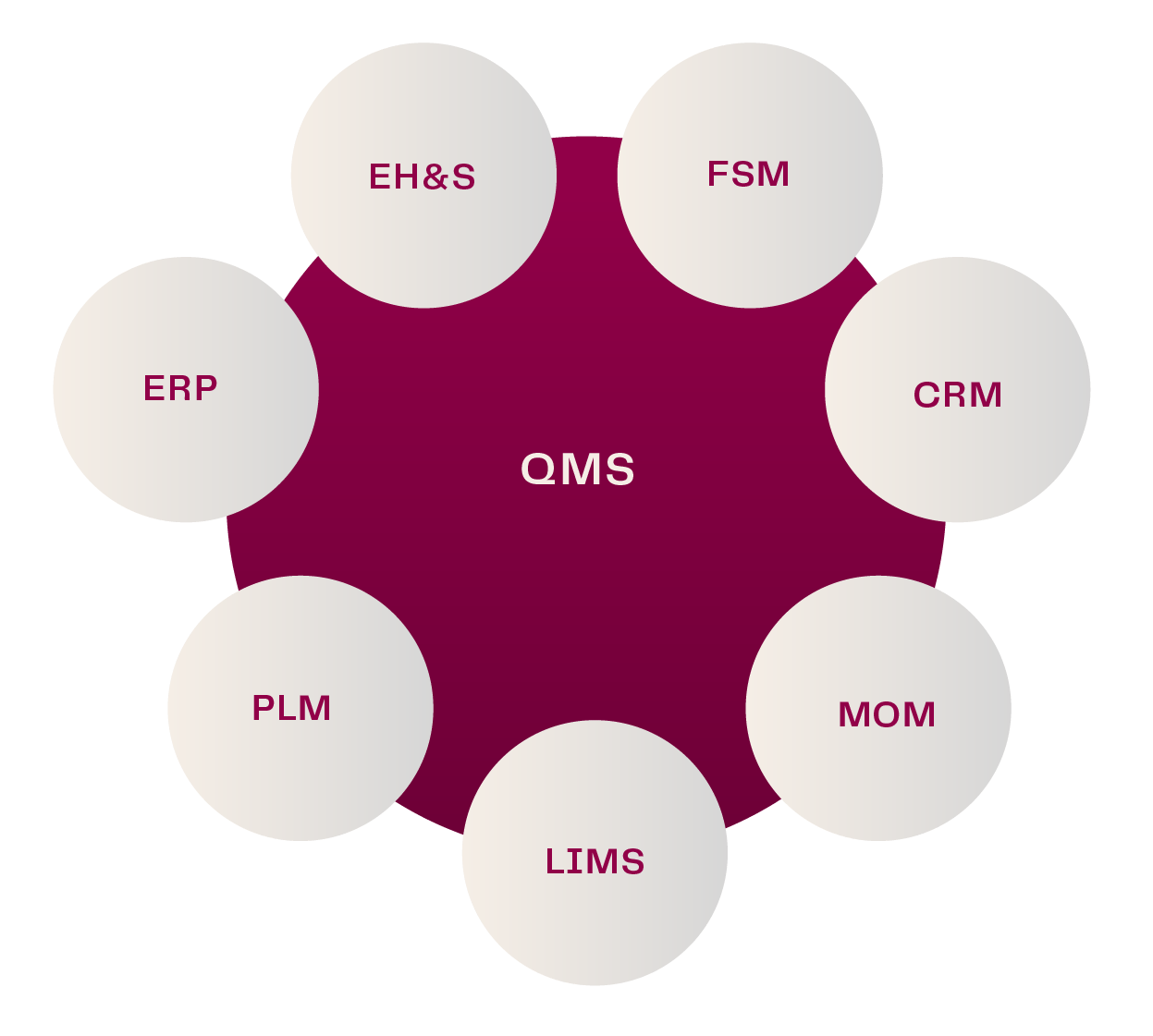
ETQ at Bio-Expo Live: Streamlining Compliance in Biopharma
Scaling compliance in biopharma is no small task. As regulations evolve and manufacturing complexities grow, companies need the right solutions to maintain quality, ensure data integrity and streamline processes.