EQMS Solutions Frequently Asked Questions
What is an EQMS?
An Electronic Quality Management System (EQMS), also known as an Enterprise Quality Management System, is defined as a software that streamlines and controls all aspects of quality, compliance, and product development within an organization. It digitizes and manages these crucial processes, ensuring uniformity and adherence to industry regulations.
How do you use an EQMS?
You can use an EQMS to assemble audit documents, alert on changes in productivity or acceptances, vet new suppliers, and more automatically. Although you can do this with a more traditional QMS, you can’t automate critical processes, incorporate advanced analytics, or respond to quality incidents in real-time.
What is an example of an EQMS?
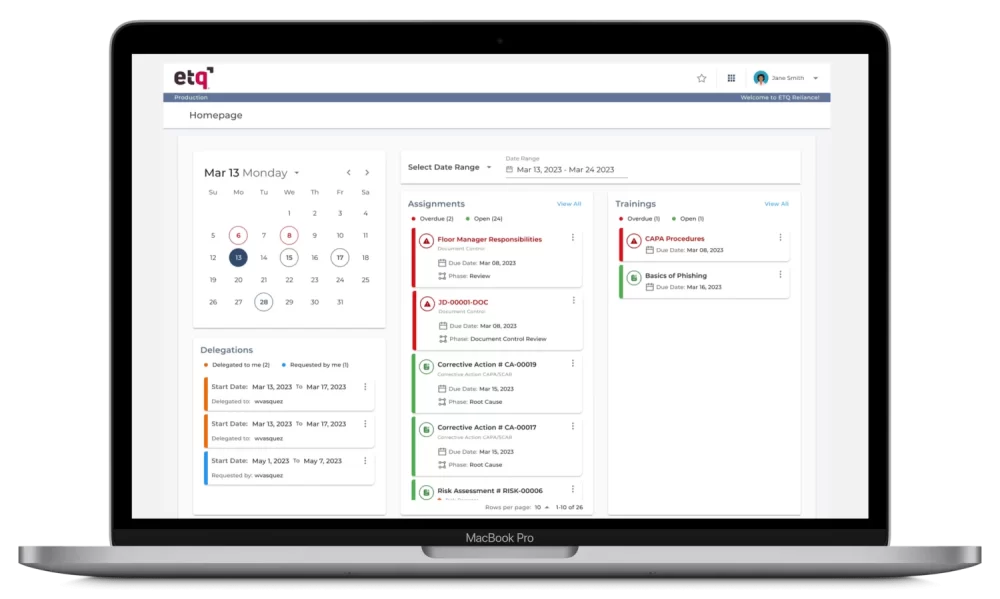
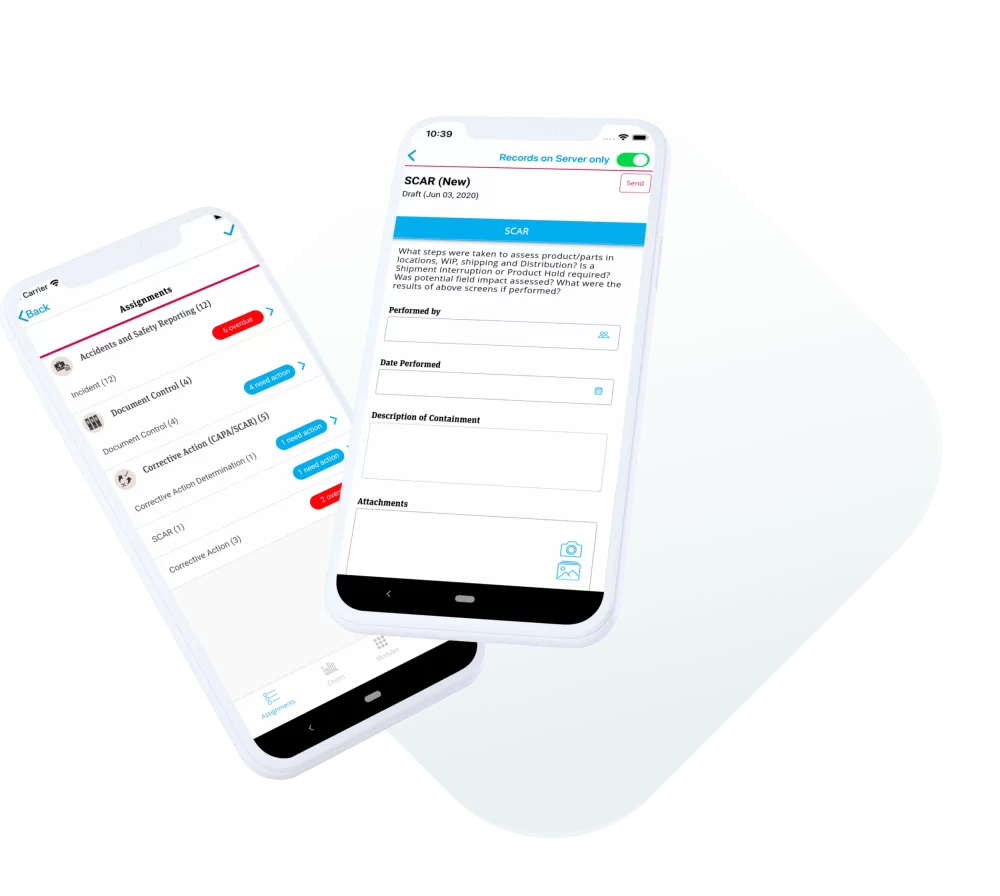
ETQ provides a robust Electronic quality management system (EQMS) as a comprehensive software solution for organizations. With features like document control, training, audit, and CAPA management, ETQ’s EQMS helps streamline and automate quality processes. The platform aims to ensure compliance and improve operational efficiency. Digitizing and centralizing quality-related data throughout the product lifecycle elevates product and process quality.
How do you validate an EQMS?
Validation is the process of confirming your EQMS meets FDA and ISO regulatory requirements, where applicable. To validate, you’ll need to understand that your servers are correctly configured, your QMS works as designed, and the product has been tested and found to perform as expected. Your vendor should be able to walk you through much of the work necessary to qualify your electronic quality management system under various guidelines.
Who needs EQMS software?
Every company with a business model that involves manufacturing or industrial processes can use an enterprise electronic quality management software. That’s because QMS is optimized towards functions such as instrumenting capital equipment, inspecting lots of finished goods, or responding to regulatory audits—functionality that forms the backbone of industrial and manufacturing organizations.
What are the types of quality management?
Quality management covers a variety of areas including document control, corrective actions, training management, risk management, nonconformance management, supplier quality, audit management and change management. An EQMS can usually handle all kinds of quality management within a single application suite. In addition, vendors can work with customers to provide custom functionality that may not exist within an out-of-the-box QMS product.
What are the benefits of using a quality management system?
Quality management systems enable higher product and service quality. This leads to fewer recalls, scrap and rework, as well as reduced warranty claims. An EQMS software that can interface with ERP, CRM, and manufacturing (MES) applications can also use the data those systems generate help users draw see “hidden” trends and patterns to make better decisions faster.
Which is better—a cloud QMS or on-premises QMS software?
Cloud QMS has several advantages over its hosted and on-premise counterparts. With cloud QMS, companies can store a nearly infinite amount of quality data with no performance slowdown. Cloud QMS implementations also enjoy higher uptime and better security. Last, cloud QMS implementations receive constant updates and support, meaning they never become obsolete.
Can I integrate this QMS software?
Depending on your QMS vendor, you can integrate with nearly any kind of application. Common choices include ERP, CRM, PLM and MES. Depending on your application stack, your QMS may natively integrate with your existing software. If not, you may have to develop an integration or ask your QMS vendor for assistance.
How much does EQMS cost?
Costs depend on the number of applications implemented and the number of users who will access your system. The advantage of deploying an EQMS in the cloud is that it will receive constant updates, be dynamically scalable and highly secure, meaning your QMS remains future proof for as long as you maintain a relationship with the vendor.
Is this quality management system right for my industry?
Many QMS solutions are specialized on a per-industry basis. Some QMS systems are built specifically for life sciences, automotive manufacturers, food and beverage producers, and more. ETQ Reliance quality management system software takes a broader approach. We’ve created a QMS system every industry can use, with specialized modules for specialized industries such as life sciences. That allows companies to cross-pollinate best practices from one industry to another, promoting a higher quality overall.
What is the difference between EQMS and QMS?
A QMS (Quality Management System) is a comprehensive framework for ensuring product or service quality, implemented manually or digitally. An EQMS (Electronic Quality Management System) specifically uses digital tools to automate and streamline quality processes, emphasizing efficiency and accessibility. The choice between them depends on an organization’s needs and desired level of automation.
What does an EQMS do?
EQMS software provides document control, audit management, nonconformance handling, training management, risk assessment, and more. EQMS enhances these capabilities by providing features such as automation, advanced analytics, and machine learning. That allows users to respond to quality events in real-time while making better decisions supported by rich data.
What should I look for in an EQMS?
When selecting an Electronic quality management system (EQMS), focus on key factors such as comprehensive modules covering document control, training, audits, and corrective actions. Prioritize a user-friendly interface for easy adoption, ensuring employees at all levels can navigate the system efficiently. Scalability is crucial to accommodate organizational growth, and the EQMS should adapt to evolving requirements.
Ensure your compliance management features meet industry standards and regulatory requirements, minimizing audit risks. Leverage integration capabilities with existing software like ERP to maintain data consistency and break down silos. Considering these factors will help you choose an EQMS that aligns with your organization’s needs and supports quality management processes.