


United Airlines Blazes New Trails as Quality Leaders

It’s Time to Take a Quality Approach to Supply Chain Disruption
Supply chain disruption is not ending anytime soon, but with the right QMS you can gain the visibility, tools, and insights you need to strengthen resiliency and mitigate many of the associated problems to keep your business moving forward.

Strategic Imperative: AMD’s Quality Journey with ETQ Reliance
High-performance computing provider AMD partnered with ETQ Reliance on its quality journey. Learn their implementation best practices.

Rheem Keeps Quality Processes Cool Amid Hot Growth
Water heater and HVAC industry leader Rheem uses ETQ Reliance to keep quality high and maintain customer satisfaction while the business grows. Read more about their quality journey.

The Growing Role of Machine Learning in Quality
In an article for Quality Magazine, ETQ CTO Morgan Palmer discusses how the future of quality will increasingly rely on artificial Intelligence (AI) and more particularly, machine learning (ML), as a way to augment the insights and capabilities of quality teams.

Build Resiliency to Mitigate Supply Chain Risk
David Issacson, ETQ’s Vice President of Product Marketing, wrote an article for SupplyChainBrain outlining strategies manufacturers can do to ensure supply chain resiliency, not only during times of disruption but in every climate.

ETQ + Hexagon: Quality Foundation for Digital Transformation
Hexagon acquired ETQ to build an integrated digital connection – connected quality -- that bridges the data gap between product quality control (QC) and in-process quality assurance (QA).
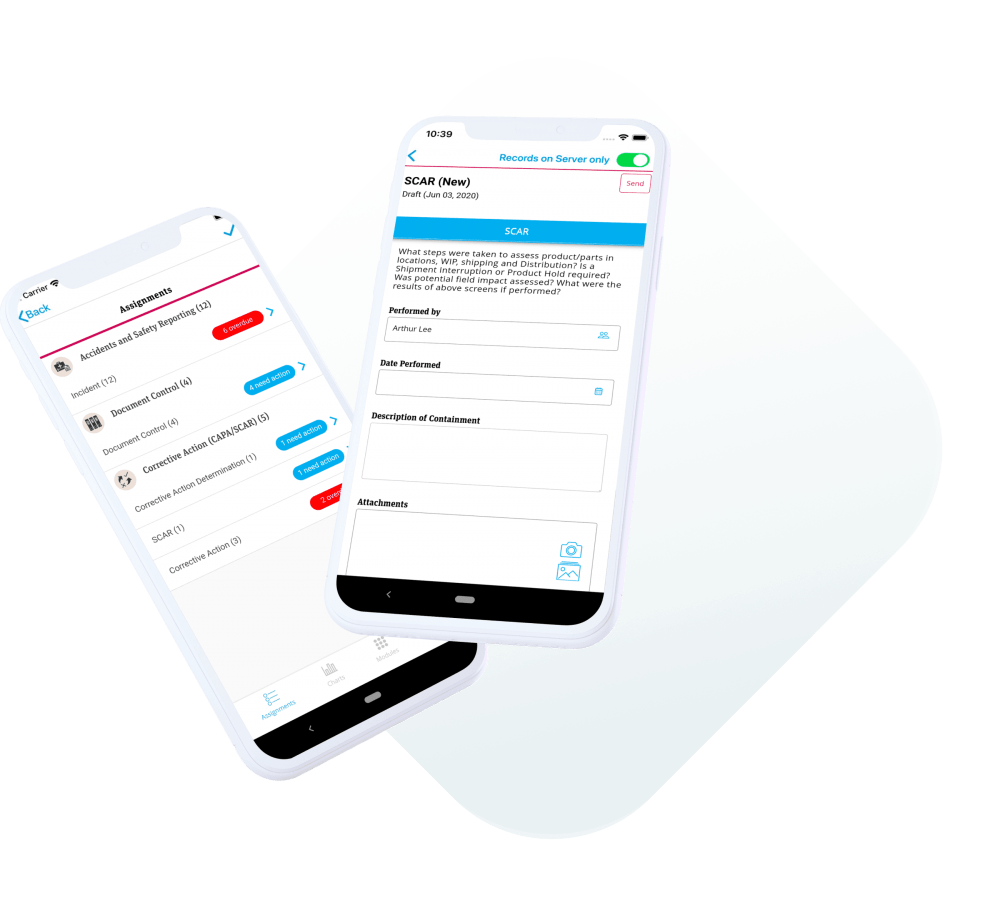
Mobile Quality Management: Solutions for Today
Introducing mobile functionality to quality management makes the process more efficient, accurate, and convenient. Learn how going mobile can be beneficial to your organization.

The Benefits of Quality Management Software – QMS
Quality management is a catalyst to organizational success that builds a pathway to product and service excellence, brand loyalty and financial improvement.

Comparing ISO 9001 & ISO 13485 Standards for Medical Devices
ISO 13485 for medical device quality management shares many similarities with ISO 9001, the leading global quality management standard with more than 1.1 million certificates worldwide.

7 Signs of a Strong Culture of Quality
Looking to build a strong quality culture? Check out these 7 signs of a culture of quality to see if you're on track.
