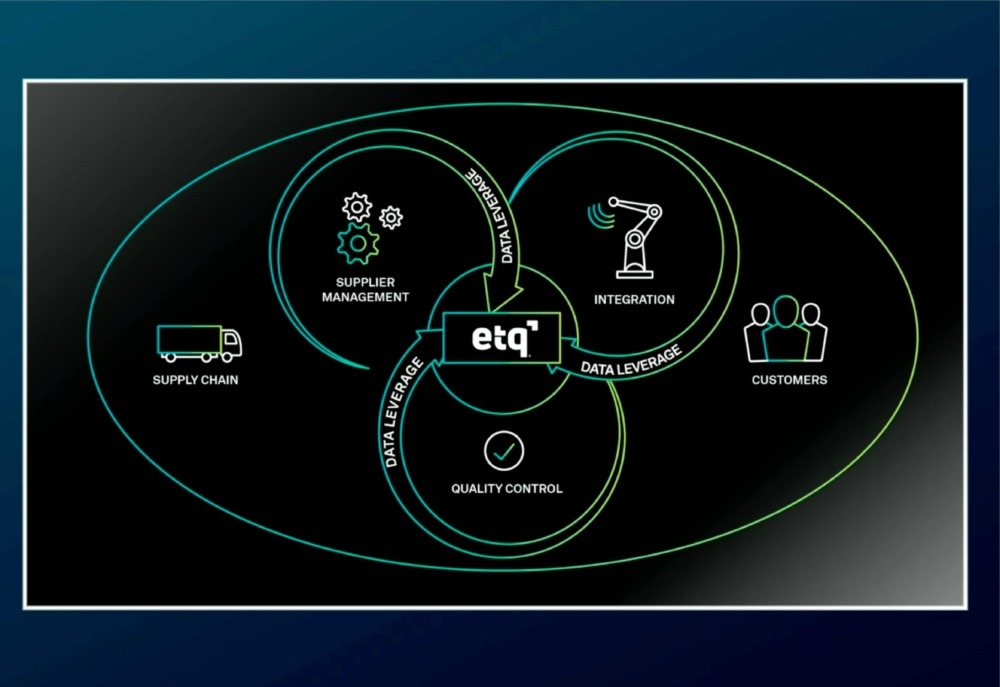
The Hidden ROI of Customer Complaint Management
In highly competitive industries, product differentiation isn’t enough to win new customers and gain market share. It’s all about the customer experience; meeting or exceeding their expectations is the new path to growth. Your product or service is irrelevant if you don’t have loyal customers.