Effective Document Control For Compliance, Quality Control and Safety
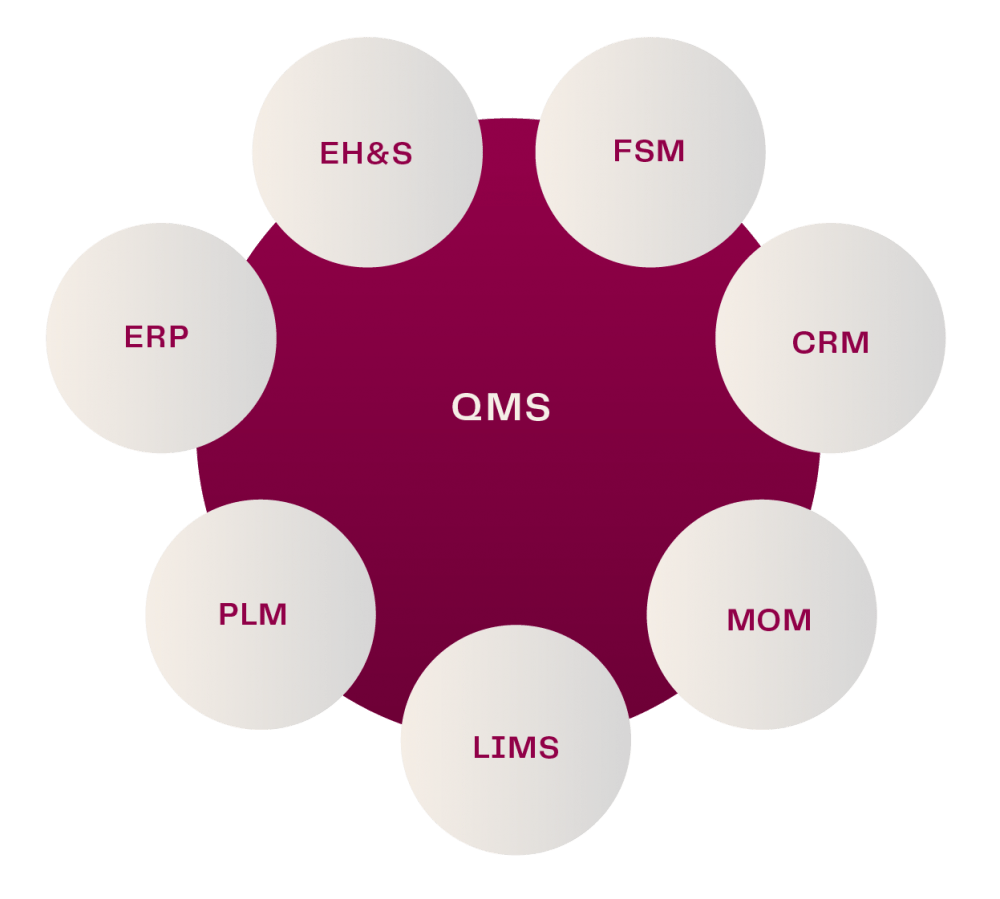
For all types of manufacturers, weakness in document control not only causes compliance issues; but it can be a major source of financial, safety and brand risk as well. Putting in place effective document control strategies should be a top priority to ensure operational excellence. This imperative was explored recently in an article in Machine Design, written by our VP, Product Marketing, David Isaacson.